
Other reported clinical experience has not identified differences in responses between the elderly and younger patients. Geriatric UseĬlinical studies of butalbital, acetaminophen and caffeine tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Safety and effectiveness in pediatric patients below the age of 12 have not been established. Because of potential for serious adverse reactions in nursing infants from butalbital, acetaminophen and caffeine, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. Nursing MothersĬaffeine, barbiturates and acetaminophen are excreted in breast milk in small amounts, but the significance of their effects on nursing infants is not known. The infant was given phenobarbital 5 mg/kg, which was tapered without further seizure or other withdrawal symptoms. Butalbital was found in the infant’s serum. Withdrawal seizures were reported in a two-day-old male infant whose mother had taken a butalbital containing drug during the last two months of pregnancy. This product should be given to a pregnant woman only when clearly needed. It is also not known whether butalbital, acetaminophen and caffeine can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. PregnancyĪnimal reproduction studies have not been conducted with this combination product. No adequate studies have been conducted in animals to determine whether acetaminophen or butalbital have a potential for carcinogenesis, mutagenesis or impairment of fertility. Carcinogenesis, Mutadenesis, Impairment of Fertility Drug/Laboratory Test InteractionsĪcetaminophen may produce false-positive test results for urinary 5-hydroxyindoleacetic acid. The CNS effects of butalbital may be enhanced by monoamine oxidase (MAO) inhibitors.īutalbital, acetaminophen and caffeine may enhance the effects of: other narcotic analgesics, alcohol, general anesthetics, tranquilizers such as chlordiazepoxide, sedative-hypnotics, or other CNS depressants, causing increased CNS depression.
#Butalbital acetaminophen caffeine serial
In patients with severe hepatic or renal disease, effects of therapy should be monitored with serial liver and/or renal function tests. Patients should take the drug only for as long as it is prescribed, in the amounts prescribed, and no more frequently than prescribed.įor information on use in geriatric patients, see PRECAUTIONS/Geriatric Use. Such tasks should be avoided while taking this product.Īlcohol and other CNS depressants may produce an additive CNS depression, when taken with this combination product, and should be avoided.īutalbital may be habit-forming. This product may impair mental and/or physical abilities required for the performance of potentially hazardous tasks such as driving a car or operating machinery. Call your doctor if you took more than the recommended dose.
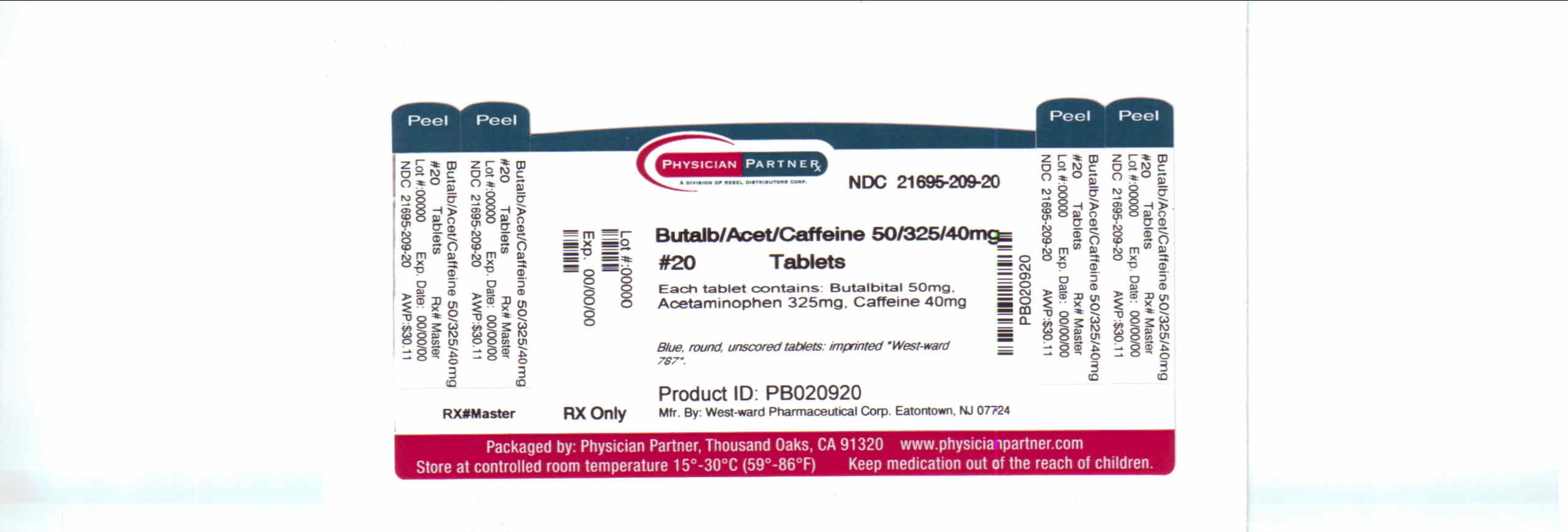
� If you develop signs of allergy such as a rash or difficulty breathing stop taking Butalbital, Acetaminophen and Caffeine Tablets, USP andĬontact your healthcare provider immediately. Do not take Butalbital, Acetaminophen and Caffeine Tablets, USP if you are allergic to any of its ingredients.

Butalbital, acetaminophen and caffeine tablets should be prescribed with caution in certain special-risk patients, such as the elderly or debilitated, and those with severe impairment of renal or hepatic function, or acute abdominal conditions.

